Sunday, October 10, 2004
6235
Impact of Vascularized Bone Marrow Transplantation Augmented with Hematopoietic Stem Cell Transplantation on Chimerism Induction Across MHC Barrier
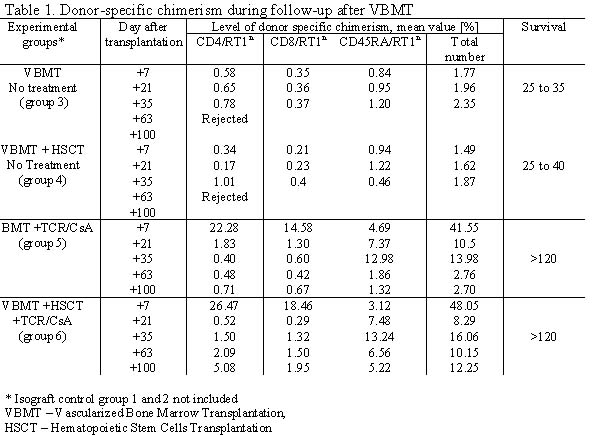
Purpose: Composite tissue allografts such as human hand comprise of vascularized bone marrow as a part of allograft transplantation. This study was designated to evaluate the efficacy of vascularized bone marrow (VBMT) and hematopoetic stem cell transplantation (HSCT) on development of chimerism and induction of donor specific tolerance. Methods: Thirty six vascularized bone marrow (femur) transplants (VBMT) containing 50x106 to 70x106 cells were performed across MHC barrier between Brown Norway (BN; RT1n) donors and Lewis (LEW; RT1l) recipients in six experimental groups of six animals each. Isograft controls between isogenic Lewis rats included group 1- VBMT and group 2 (VBMT + HSCT) without treatment. Rejection controls between BN and LEW rats included group 3 – VBMT and group 4 (VBMT+HSCT) without treatment. In treatment groups across MHC barrier (BN to LEW) group 5 – VBMT and group 6 (VBMT + HSCT), allograft recipients were subjected to 7 day protocol of abTCRmAb/CsA therapy. Intraosseouss HSC transplant (35x106 cells) into the contralateral femur was performed in groups 2, 4 and 6. Flow cytometry was used for evaluation of efficacy of immunomodulation and donor specific chimerism (MHC class I - RTIn) at post-transplant days 7, 21,35, 63 and 100. Results: Isograft controls (group 1 and 2) survived over 120 days. Non-treatment allografts showed signs of rejection (bone exposure, inflammation, infection and hair loss) between days 25 and 35 post-transplant in group 3 (VBMT), and between days 25 and 40 post-transplantat in group 4 (VBMT+HSCT). Allograft recipients in treatment groups (groups 5 and 6) survived with no sign of rejection throughout the follow-up period up to 120 days. Transplantation of VBM (group 3) and VBM+HSC (group 4) without treatment resulted in development of low level donor-specific chimerism between days 7 and 35 in both T and B- lymphocyte cell populations and ranged from 1.49% to 2.35% (Table 1). In groups 5 and 6 (abTCRmAb + CSA protocol) flow cytometry at day 7 revealed >95% efficacy for T-lymphocyte depletion. T-lymphocytes repopulated to the pretreatment level at day 63 post-transplantat. At day 7 after transplantation high level of donor specific chimerism was observed in group 5 for RT1n/CD4+ (22.28%) and for RT1n/CD8+ (14.58%) of T cell subpopulations. In group 6 augmented with HSCT highest level of multilineage chimerism was seen at day 7 and revealed 26.47% of RT1n/CD4+ and 18.46% of RT1n/CD8+. In both abTCRmAb/CSA treatment groups T-lymphocyte chimerism decreased over time and switched to predominantly B-lymphocytes chimerism at day 21 post-transplant (Table 1). In group 5 chimerism was maintained through the B-lymphocytes, whereas in group 6 augmented with HSCT chimerism was maintained through both T- and B-lymphocytes, with B-lymphocytes playing most important role in the chimerism maintenance (Table 1). Conclusions: Transplantation of vascularized bone without immunosuppression provides a substantial source of bone marrow derived hematopoietic cells within its natural microenvironment, leading to development of donor specific chimerism up to 35 days post-transplant. Therapy with 7 day abTCR/CsA protocol facilitates development and maintenance of stable mixed multilineage chimerism up to 100 days post-transplant leading to tolerance induction in fully MHC mismatched transplants. Augmentation of vascularized bone transplant with direct intraosseous HSC transplantation allows for chimerism maintenance at 75% higher level up to 100 days post-transplant.
View Synopsis (.doc format, 110.0 kb)